A fish story: How does your brain work? Ask a minnow
It may be hard to believe, but those ubiquitous striped minnows swimming in aquariums may hold secrets to the human brain, according to Assistant Professor Justin Kenney. He is using the little fish, known as zebrafish (Danio rerio), to gain a fundamental understanding of how the human brain's complex network controls behavior and possibly point the way to new treatments for psychiatric disorders.
"This is an interesting and challenging area, because at the moment, we don't have a lot of good drugs to give people for anxiety, depression, or any of these sorts of disorders and a big part of that is there is a lot we don't know about how the brain works and how these disorders are related to brain activity," Kenney explained.
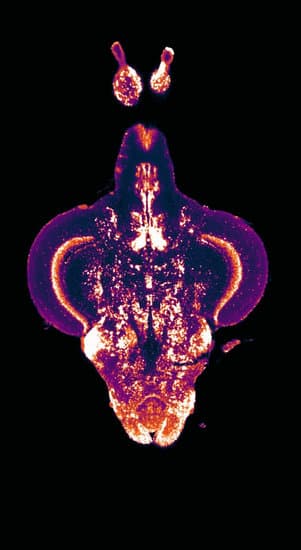
He sees zebrafish as a good model organism to bridge that gap for several reasons. For one thing, they may be simple organisms compared to humans, but their brains control behavior in the same basic ways, he says. In addition, zebrafish are very prolific, with a female laying 50-100 eggs at a time and those young can begin breeding when they are only 10-12 weeks old. This gives researchers plenty of opportunities to genetically modify the eggs, which can reveal the role of specific genes in behavior. "That means we can generate a lot of observations per manipulation, which in turn yields the large amounts of data required for some of the computational tools, machine learning and big-data analytics, that are coming online now," he said.
Casting the net
Kenney and his group are using several approaches to study brain-behavior relationships. In one, they monitor the anxiety-related behaviors fish exhibit when they are placed into unfamiliar tanks. These include preferentially swimming at the bottom of a tank rather than the top and at its periphery rather than in the center. Using a new stereoscopic-imaging camera that is able to track the fish's exact three-dimensional movements in a novel tank, the researchers hope to identify the specific molecular pathways that turn off or on genes, or otherwise exacerbate or dampen anxiety.
Kenney and his group are also studying brain-behavior relationships by using the state-of-the-art techniques of tissue clearing and light-sheet microscopy to not only image the whole brain at once but also to identify which regions of the brain are interacting at any given time. "With these technologies, we should be able to image an entire zebrafish brain at micron resolution in about an hour, which means it is now feasible to generate the massive amounts of data needed to do the types of analyses required to study brain networks," he said.
In addition, they are exploring associative learning and memory. This involves introducing a fish to a tank, letting it get used to the new environment and then reintroducing it to the tank a day later, but dispensing a short series of mild shocks to the water. This results in a characteristic behavioral response:

The fish reduces its swimming activity. Although the shocks are only administered on a single occasion, the fish remembers that bad experience and continues to exhibit the reduction in swimming activity when it is reintroduced to that tank hours and even days later. Using this experimental design, researchers can study the genetics of memory loss and the effects of memory-affecting drugs. The central question is whether interfering with gene transcription or disrupting other molecular pathways results in similar brain network activation profiles. "In other words," he described, "how stereotyped is the brain activity pattern that underlies a given behavior? Does it change depending on the history or genetic background of the animal, or is it the same no matter what?"
Reeling it in

Kenney and his research group are especially interested in the brain networks of fish that are naturally more anxious than others. "That will help us figure out what's being engaged differently in these two groups of fish," he said. It will also be useful in learning how anti-anxiety drugs work. "In other words, are we actually moving these anxious animals into a state that looks like a naturally non-anxious animal, or are we moving them to some third state that's completely different from anything else that we've seen?" He remarked, "My hypothesis is that even though the animal is exhibiting the same low-anxiety behavior, the brain network activation profiles between naturally low-anxiety animals and those that are drugged and also between those receiving one drug vs. another, are actually going to be different."

The ultimate goal, he said, is to be able to identify which drugs stimulate brain network activity profiles that most closely mimic those seen in naturally low-anxiety animals, because such drugs will probably have fewer of the side effects, such as nausea, dizziness and lethargy, that are common to many currently used anxiety medications.
"If you think about it, there really hasn't been much improvement in drugs for treating psychiatric disorders in the past 20 years, mainly because we just don't understand enough about the brain and how it works to even start to ask the right questions," he contended. "This is why it's important to investigate the general principles of brain function, especially at the network level and dovetail it into the context of a psychiatric disease."
Kenney added, "With this work, I can satisfy both the parts of myself - the side that is curiosity-driven and the side that wants to see how our work can improve the human condition."